Supercapacitors, also known as supercapacitors or electrochemical double-layer capacitors (EDLCs), are a new type of energy storage device that realises energy storage through the electrostatic charge accumulation of electrode materials and electrolyte interfaces. The core advantages of its high power density and fast charging and discharge cycle depend on the specific surface area and conductivity of the electrode material – and activated carbon, especially powdered activated carbon, is particularly prominent in these two aspects. With its unique structure and chemical properties, activated carbon has become an indispensable core material for optimising supercapacitor capacitance, power density, durability and commercial feasibility, and plays an irreplaceable role in the research and development and application of supercapacitors.
Supercapacitors, also known as supercapacitors and electrochemical double-layer capacitors (EDLCs), its energy storage principle is different from that of traditional capacitors. The core is to realise energy storage through the electrostatic charge accumulation of the electrode and the electrolyte interface, not the chemical reaction. Compared with batteries, the most significant advantages of supercapacitors are high power density and extremely fast charging and discharge speed, which can complete charging and discharge in a short time and have a longer cycle life.
Electrode material is the core factor that determines the performance of supercapacitors, in which the specific surface area and conductivity of the electrode material directly affects its energy storage capacity and charging and discharging efficiency: the larger the specific surface area, the more charge adsorption sites, the higher the energy storage capacity; the better the conductivity, the lower the resistance to electron migration, the faster the charging and discharging speed. As a commonly used electrode material, activated carbon has a very high specific surface area (usually more than 1500 square meters/g) due to its porous structure, which makes it the preferred electrode material for supercapacitors.
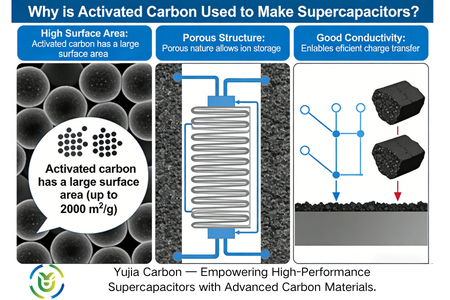
The specific surface area is one of the core advantages of activated carbon, which directly determines the adsorption capacity of the material – the larger the specific surface area, the more ions and molecules can be adsorbed. In supercapacitors, activated carbon, as an electrode material, provides sufficient space for charge separation and storage, thus significantly improving the capacitance value of the capacitor. The specific surface area of activated carbon used for supercapacitors is usually between 1000-3000 square metres/gram, which provides a solid foundation for charge storage.
As an electrode material, electrical conductivity is the key to ensuring the smooth migration of electrons. Activated carbon itself has low resistance and electron migration resistance, which can effectively reduce the internal loss of supercapacitors, accelerate the charging and discharge speed, and improve the energy conversion efficiency. This feature enables supercapacitors to quickly respond to charging and discharge needs and adapt to high-power output application scenarios.
Supercapacitors need to go through thousands of charging and discharge cycles in practical applications, so electrode materials must have good durability and stability. With its unique porous structure and chemical inertness, activated carbon has excellent corrosion resistance and structural stability. It can maintain stable performance during long-term use and is not prone to structural degradation, thus effectively prolonging the service life of supercapacitors and improving the reliability of its long-term operation.
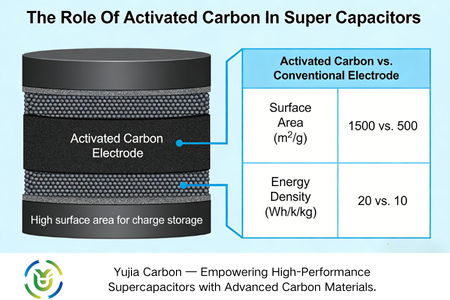
One of the most prominent features of activated carbon is its extremely high specific surface area. The specific surface area of some high-quality products can exceed 1,500 square metres/gram. This huge surface area provides a large number of active sites for charge accumulation, and the charge storage of supercapacitors happens at the interface between the electrode and the electrolyte. Therefore, the high specific surface area of activated carbon electrodes can adsorb more electrolyte ions, directly improve the capacitance capacity of the super capacitor, and enhance its energy storage capacity.
Activated carbon has a graded porous structure, which is mainly divided into micropores (less than 2 nanometers), medium pores (2-50 nanometers) and large pores (greater than 50 nanometers). Pores of different pore sizes have different functions. Among them, micropores are the main place of ion adsorption, which can increase the charge storage point and improve the capacitance value; medium pores and large pores act as ion transmission channels, accelerating the migration speed of ions during charging and discharging and reducing the resistance of ion transmission. This balanced aperture distribution can optimise the energy density and power density of supercapacitors at the same time, and realise the double improvement of energy storage capacity and charging and discharging speed.
The charge storage of activated carbon electrodes mainly relies on the physical adsorption mechanism, which is an illegal process – that is, the ions in the electrolyte form electrochemical layers on the surface of the activated carbon electrode, and the whole process does not involve chemical reactions. The advantage of this physical adsorption mechanism is that the adsorption and desorption process of ions is reversible and fast, without complex chemical reactions, thus giving supercapacitors extremely high power density and ultra-long cycle life, avoiding material loss caused by chemical reactions.
At the interface between the activated carbon electrode and the electrolyte, a layer of electrochemical double layer (EDL) will be formed: the positive and negative ions in the electrolyte will be arranged on both sides of the interface, only a few E (1 E = 0.1 nanometers) between them. According to the capacitance calculation formula C = k × A / d (where C is the capacitance, k is the dielectric constant of the dielectric, A is the electrode surface area, and d is the double layer spacing), the capacitance value is directly proportional to the electrode surface area and inversely proportional to the double electric layer spacing. The high specific surface area and porous structure of activated carbon can maximise the surface area of the electrode (A) and shorten the distance between the ion and the electrode surface (d), thus significantly improving the capacitance value of the supercapacitor.
The pore structure of activated carbon directly affects the capacitance and power density of the supercapacitor: the larger the number of micropores, the more charge adsorption points, and the higher the capacitance value; the better the smoothness of the medium and large pores, the faster the ion migration speed, and the higher the power density. Therefore, balanced pore size distribution is the key to activated carbon electrodes – it can not only improve the energy storage capacity through micropores, but also ensure rapid charging and discharging through medium and large pores, so as to achieve both high energy density and high power density and meet the needs of different application scenarios.
The specific surface area is an important basis for the energy storage capacity of electrode materials. The difference between the three is obvious: the specific surface area of activated carbon is 1000-3000 square metres/gram, which is superior to carbon nanotubes (1500-2000 square metres/gram), and is at the same level as graphene (2000-2600 square metres/gram), providing sufficient active sites for charge storage.
The conductivity directly affects the charging and discharge speed of the super capacitor. The conductivity hierarchy of the three is clear: carbon nanotubes have the highest conductivity, followed by graphene, and the conductivity of activated carbon is moderate, but it can meet the regular use needs of commercial super capacitors and effectively reduce the internal electron migration resistance.
Cost is the key to the large-scale commercial application of materials, and the cost gap between the three is significant: activated carbon has an absolute cost advantage, and the price is much lower than that of graphene and carbon nanotubes, while the latter two are expensive due to the complex preparation process, which limits their large-scale promotion.
Although graphene and carbon nanotubes are superior to activated carbon in some properties, their high cost and complex preparation process limit their large-scale commercial applications. With the advantages of low cost, simple preparation and balanced performance, activated carbon has become the most practical and mainstream electrode material for commercial super capacitors at present.
|
Electrode Materials |
Specific surface area (m2/g) |
Conductivity |
cost |
Cycle life |
|
Activated Carbon |
1000-3000 |
Medium |
Low |
Very high |
|
Graphene |
2000-2600 |
High |
High |
High |
|
Carbon Nanotubes |
1500-2000 |
Very high |
Very high |
High |

Electrode materials are the core components of supercapacitors, and their performance and cost directly determine the overall quality of supercapacitors, while powder activated carbon is one of the most widely used electrode materials at present. Activated carbon is a porous carbon material formed through chemical or physical treatment. Its unique porous structure creates a large number of sites where electrochemical reactions can take place, enabling efficient storage and release of electrical energy.
The advantages of activated carbon powder are outstanding: on the one hand, its high specific surface area enhances the contact efficiency between electrolyte ions and the electrode surface, thus increasing the capacitance value and lowering the internal resistance; on the other hand, its porous structure allows for the reversible adsorption and detachment of ions, and possesses excellent stability and durability, which significantly extends the cycle life of supercapacitors. Based on these advantages, activated carbon based supercapacitors perform well in scenarios that require high long-term reliability, such as renewable energy storage and backup power systems.
Chemical activation method mainly treats carbon materials with chemical reagents (e.g., potassium hydroxide, phosphoric acid, etc.). The core advantage of this method is high preparation efficiency, which can quickly prepare activated carbon with large specific surface area and uniform pore size distribution, and it is suitable for scenarios that require high performance of activated carbon and pursue preparation efficiency.
Physical activation achieves the construction of porous structure of activated carbon by means of high-temperature oxidation (e.g., water vapour, carbon dioxide activation). Compared with chemical activation, the preparation process is more environmentally friendly, with no chemical reagent residue, but it takes a relatively long time, which is suitable for scenarios with higher requirements for environmental protection and no urgency for mass production.
In terms of pore structure, the ratio of micropores, mesopores and macropores can be optimised by adjusting the parameters of the preparation process to enhance the efficiency of ion adsorption and transport. Reasonable regulation of the proportion of the distribution of the three types of pores can allow the micropores to give full play to ion adsorption, while keeping the mesopores and macropores open to reduce the resistance during ion migration. This optimisation further balances the capacitance capacity of the supercapacitor with the charging and discharging speeds, making the electrode performance more suitable for practical applications.
In terms of conductivity, the electrode resistance can be reduced by doping with conductive materials such as graphene and carbon nanotubes. Graphene and carbon nanotubes have excellent conductivity, and reasonable doping with activated carbon can build a smoother electron transport channel and reduce the loss of electron migration process. This optimisation not only improves the charging and discharging efficiency of the supercapacitor, but also further enhances the overall conductivity stability of the electrodes, which is suitable for high power output scenarios.
In terms of stability, surface modification can be used to enhance the corrosion resistance and structural stability of activated carbon. Surface modification can form a protective film on the surface of the activated carbon, effectively resisting the corrosion of the electrolyte and avoiding structural degradation of the electrode material. At the same time, the modification treatment can also enhance the compatibility between the activated carbon and the electrolyte, so that the electrode maintains stable performance in long-term charge and discharge cycles, further extending the service life of supercapacitors.

Supercapacitors can be used in various types of consumer electronic products such as smart phones, wearable devices, power tools, etc., often as an auxiliary power supply or backup power supply. It can effectively solve the pain point of slow charging and discharging speed and short cycle life of traditional batteries, and quickly provide emergency power in case of sudden power failure of equipment. Its compact size and stable performance can also adapt to the development needs of consumer electronics miniaturisation and lightweight, further enhancing the user experience.
In the field of new energy, supercapacitors containing activated carbon have a wide range of application prospects, and can be used in the storage of renewable energy sources such as solar energy and wind energy. It can effectively balance the fluctuation of new energy supply, store excess electricity and release it when there is a shortage of energy, so as to guarantee the stability of energy supply. It can also be applied to hybrid vehicles, assisting the power battery to achieve fast starting and braking energy recovery, which not only reduces energy consumption, but also significantly improves the fuel economy and range stability of the car.
In the field of industrial production, the role of supercapacitors is also indispensable, widely used in power grid stability, industrial equipment backup power and other scenarios. It can quickly respond to voltage fluctuations in the power grid, play a role in adjusting peaks and filling in valleys, stabilising the frequency of the power grid, and guaranteeing the safe operation of the power grid. As a backup power supply for industrial equipment, it can start instantly when there is a sudden power outage to provide emergency power for the equipment, avoiding production interruptions and equipment damage due to sudden power outages and reducing economic losses.
Although activated carbon based supercapacitors have many advantages, there are still some limitations: first, the energy density is relatively low, compared with lithium batteries, there is still a gap in its energy storage capacity, and it is difficult to meet the needs of long range scenarios; second, the cost of the material is still optimisation space, although the cost of activated carbon itself is relatively low, but the complexity of the preparation process of high-performance activated carbon leads to high overall electrode costs, which limits its use in some high-end application in some high-end scenarios.
In view of the current limitations, future research on the application of activated carbon in supercapacitors mainly focuses on two directions: firstly, composite material research and development, composite activated carbon with graphene, carbon nanotubes, metal oxides and other materials, combining the advantages of each material, and at the same time improving the energy density and power density of supercapacitors; secondly, process optimization, by improving the preparation of activated carbon and electrode molding process, reducing production costs and improving the consistency and stability of the product. cost, enhance the consistency and stability of the product, and promote its large-scale commercial application.
Activated carbon has become the core material of supercapacitor electrodes by virtue of its high specific surface area, good electrical conductivity, excellent stability and cost advantages, and plays an irreplaceable role in charge storage and ion transport. Compared with graphene and carbon nanotubes, it is more cost-effective and more suitable for large-scale commercialisation, and is the core support for industrial development. In the future, through composite modification and process optimisation, it is expected to break through the limitations of energy density, play a more important role in new energy storage, electric vehicles and other fields, and promote the green and efficient development of the energy storage industry.